Choosing the Best pH Electrode for Your Application
Finding the right electrode for a specific application might seem like a mind-boggling exercise! By following these charts, you can find the right electrode and also understand the reasons why one type is better than another.
Learn MoreElectrode Comparison Charts
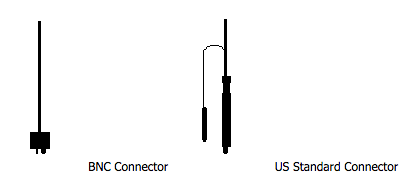
1. BNC Connector* VS. US Standard Connector*
- Smaller (fits better on specially smaller or hand held meters).
- Inexpensive (BNC connectors are widely used on TV sets).
- Fastens better (due to the twisting mechanism).
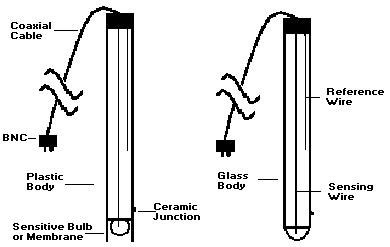
*Connectors: Before looking at different types of electrodes, ensure that the male connector at the end of the electrode cable matches the female connector on the pH meter. The vast majority of modern pH meters take BNC. (see Figure 1)
2. Glass Body Electrodes* VS. Epoxy (Plastic) Body Electrodes*
- Slightly faster (due to larger surface area of the bulb).
- Easier to clean (larger bulb surface area).
- Available in more shapes for special applications.
- Stands higher temperatures (typically up to 100 Centigrade; plastic only up to 80 °C).
- More rugged (the electrode stem and bulb are well protected).
- Costs less than glass electrodes.
3. Conic-Shaped Tip* VS. Sphere-Shaped Tip*
- Easier to clean and maintain. Sample trickles down to the tip. Best for applications such as dairy.
- The tip is more rugged, hence better for penetration.
- Can last longer.
- A few seconds faster due to the larger surface area of the bulb.
- Normally costs less.
*Conic/sphere shaped refers to the shape of the membrane only (see Figure 2).
4. Gel-Filled Electrodes* VS. Refillable Electrodes*
- Requires less maintenance. No refilling required.
- Resists higher pressure (+15-30 psi).
- Electrode could last longer if electrolyte is changed.
- A different electrolyte, for example, silver-free or calomel for special applications can be used.
- Slightly faster response due to greater flow of electrolyte into the sample.
*Gel-filled/Refillable refers to the kind of electrolyte inside the reference chamber.
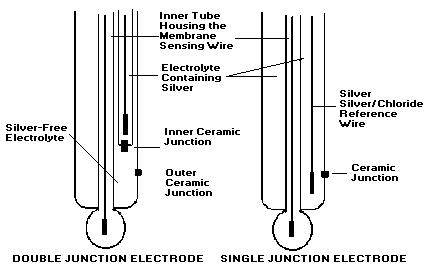
5. Double Junction Reference* VS. Single Junction Reference*
- Protects the sample from having any silver contamination from the electrolyte.
- Almost no silver is deposited on outer ceramic as electrolyte is lost into sample hence no clogging from inside.
- Protects the Silver wire from being contaminated due to prolonged use, or in highly acidic or alkaline samples.
- Normally costs less. (The Hanna Double Junction Plastic Electrodes are however supplied at the same price as standard electrodes).
- Longer life.
6. Triple Ceramic Junction* VS. Single Ceramic Junction
- Allow a higher outflow of electrolyte, hence less likely to clog.
- Recommended for low conductivity samples due to higher outflow of KCl.
- Slightly faster response.
-
Costs less.
*Triple Ceramic have 3 Ceramics as opposed to a single outer ceramic on the stem of elec.
7. Ceramic Junction VS. Open Junction*
- Lasts longer.
- Costs less.
- Stands higher temperatures (Typically 80°C or higher whereas open junction max. Temp. Is around 50°C)
- Stands higher pressure.
- No clogging (with a conic membrane, this becomes the ideal electrode for food samples with protein or glucose content or any medium which clog normal electrodes)
- Faster.
*Open Junction means that there is no ceramic separating the sample from the electrolyte.